2nd Annual Genomics Meeting: Genomics and Translational Medicine in Myeloma

The IMS Genomics Meeting was developed in response to the IMS-FDA joint workshop to further consider essential needs in myeloma. The initial invitation-only meeting in 2023 provided an opportunity for myeloma experts from around the world to develop a consensus to define ‘high-risk multiple myeloma’. Building upon this success, the Second IMS Genomics Meeting will be a public meeting led by global myeloma KOL.
In addition to which, this day and a half-long meeting will include sessions on:
- Risk Stratification validation
- Genomically targeted therapies
- New technologies
- Genomic Biomarkers for stratification of clinical studies
- New clinical trials for patients with HR disease
Our sponsors:
Gold

Silver

Tentative Agenda
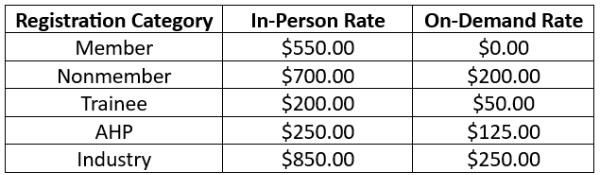
In Person Registration
- Admission to scientific sessions.
- Admission to breakfasts, lunches, and coffee breaks.
- Certificate of attendance.
- One-year access to on-demand session recordings. Access instructions will be provided 3 weeks post-event.
On-demand Registration
- One-year access to on-demand session recordings. Access instructions will be provided within 3 weeks post-event.

